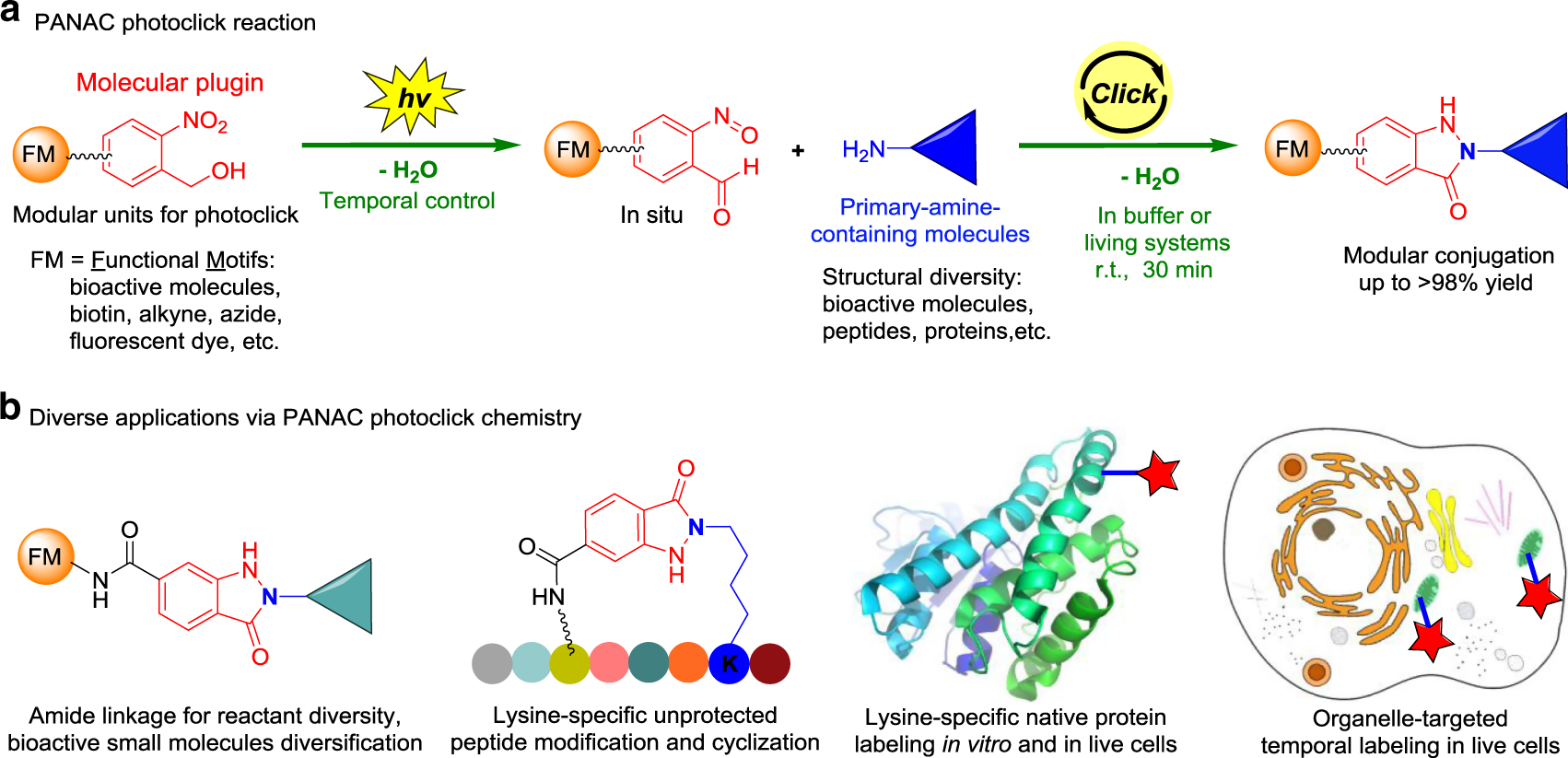
Light-induced primary amines and o-nitrobenzyl alcohols cyclization as a versatile photoclick reaction for modular conjugation | Nature Communications

Monobodies as tool biologics for accelerating target validation and druggable site discovery - RSC Medicinal Chemistry (RSC Publishing) DOI:10.1039/D1MD00188D

n→π* Interactions of Amides and Thioamides: Implications for Protein Stability | Journal of the American Chemical Society
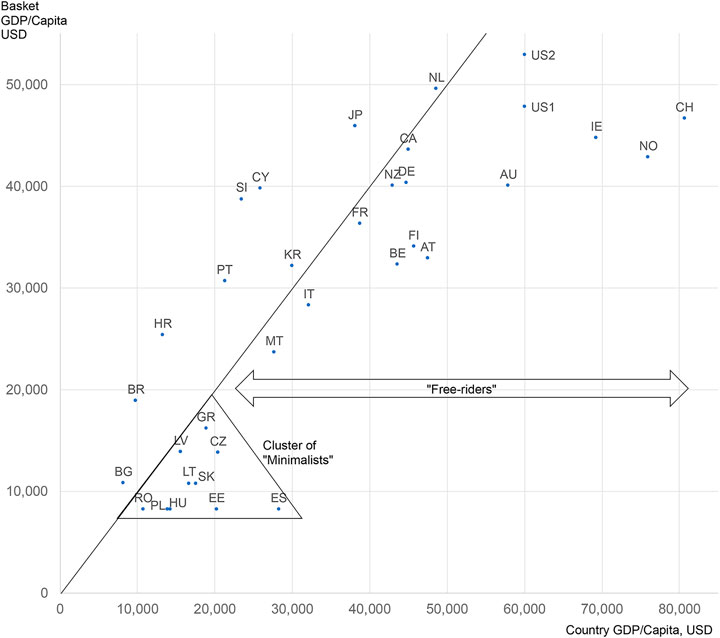
Frontiers | Assessing the Consequences of External Reference Pricing for Global Access to Medicines and Innovation: Economic Analysis and Policy Implications