The rate constant of a reaction is 0.0693 min^(-1). Starting with 10 mol, the rate of the reaction after 10 min is

The energy of activation and specific rate constant for a first order reaction at 25^∘ C are 100kJ/mole and 3.46 × 10^-5sec^-1 respectively. Determine the temperature at which half - life of

Calculate the half life of a first order reaction from their rate constants given below:(a) 200 s^-1 ; (b) 2 min^-1 ; (c) 4 year^-1 .

If the half life period for a first order reaction is 69*3 seconds, what is the value of its rate constant ?
10. rate constant of first order reaction is 0.0693per minute . calculate the percentage of reaction remaining at the end of the 60 minute.(with detail solution )

For a reaction A ⟶ B + C . it was found that at the end of 10 minutes from the start the total optical rotation of the system was 50^o and

SOLVED: What is the rate constant of a first-order reaction that takes 456 seconds for the reactant concentration to drop to half of its initial value?

The unit of first order rate constant when concentration is measured in terms of pressure and time - YouTube

Half life of a first order reaction is 2.1xx10^(12)s. Calculate the rate constant of the reactio... - YouTube

At a certain temperature, the rate constant of a first - order reaction is 1.40 min ^-1 . Find its half - life.

The reaction `A(g)toB(g)+2C(g)` is a first order reaction with rate constant `2.772xx10^(-3)sec^(-1) - YouTube

Calculate the half life of the first order reaction from their rate constant given as a) 200s^(-) b) 2min^(-1) c) 4 "year"^(-1).

A first order reaction is found to have a rate constant k= 5.5 xx 10^(-14)s^(-1). Find half-life of the reaction.

The reaction is given below, the rate constant for disappearance of A is 7.48 × 10^-3sec^-1 . The time required for the total pressure in a system containing A at an initial
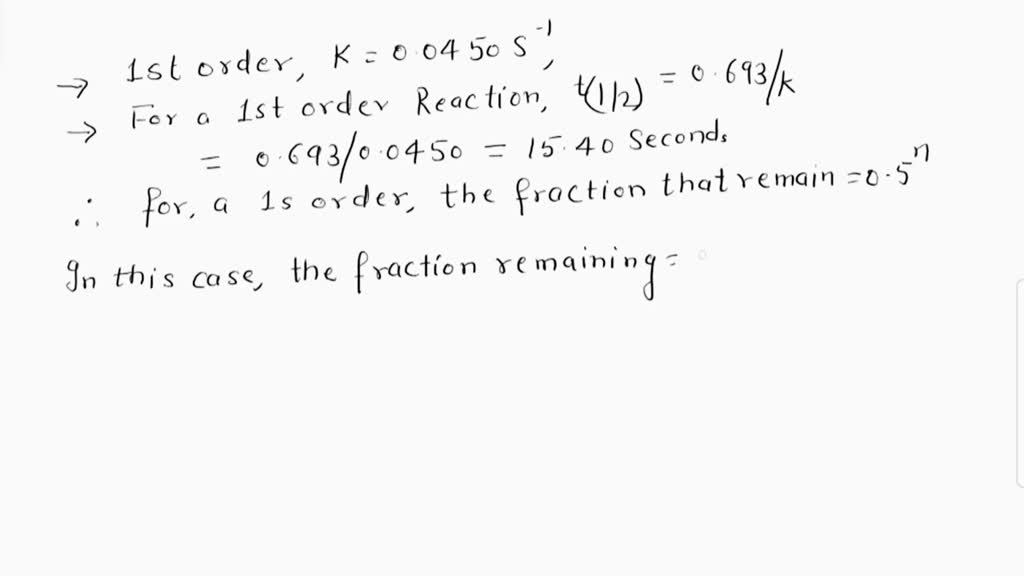
SOLVED: If a reaction is first order with a rate constant of 0.0450 s⁻¹, how much time is required for 65% of the initial quantity of reactant to be consumed?