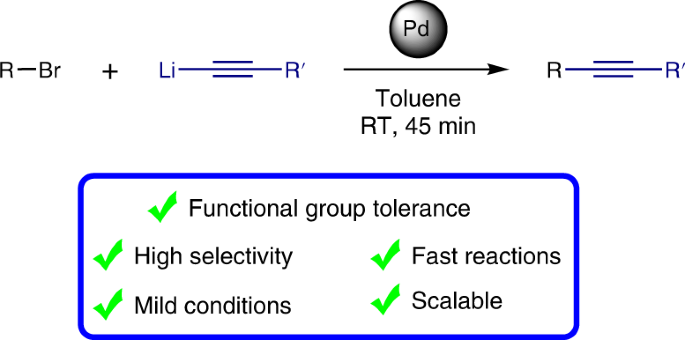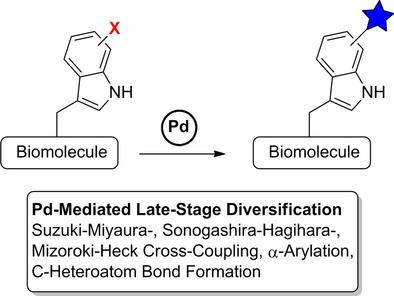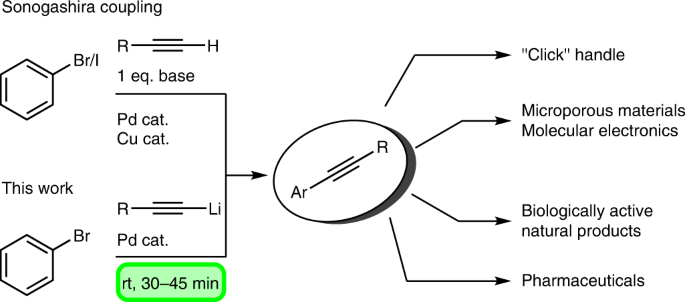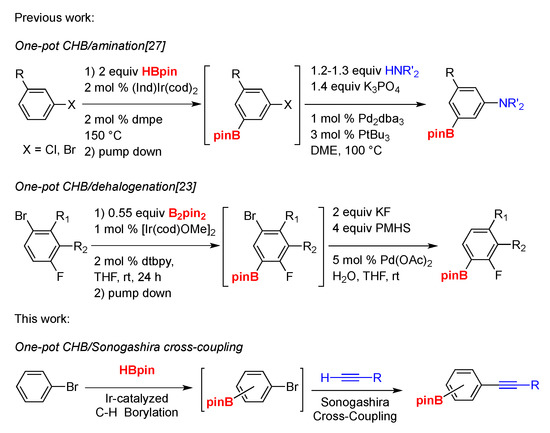
Molecules | Free Full-Text | One-Pot Iridium Catalyzed C–H Borylation/ Sonogashira Cross-Coupling: Access to Borylated Aryl Alkynes | HTML

Efficient and Copper‐Free Sonogashira Cross‐Coupling Reaction Catalyzed by Pd(OAc)2/Pyrimidines Catalytic System - Li - 2005 - European Journal of Organic Chemistry - Wiley Online Library

Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis. - Abstract - Europe PMC

Recent advances in bioorthogonal reactions for site-specific protein labeling and engineering - ScienceDirect
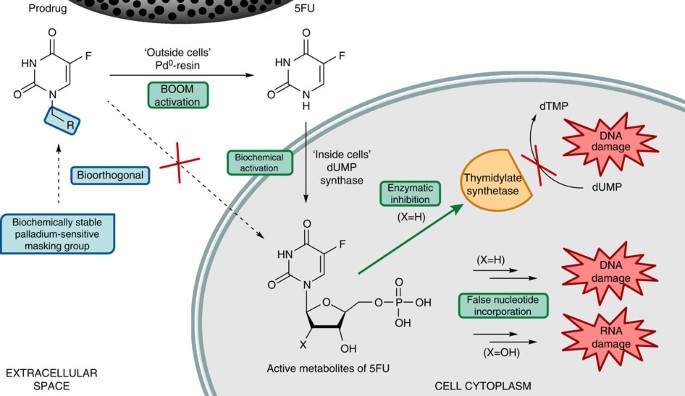
Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach | Nature Communications
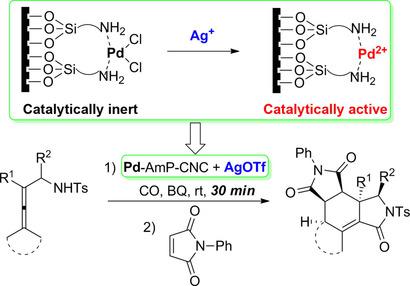
Silver-Triggered Activity of a Heterogeneous Palladium Catalyst in Oxidative Carbonylation Reactions. - Angew. Chem. Int. Ed. - X-MOL
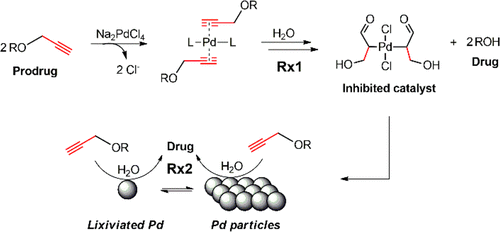
Mechanism of Palladium(II)-Mediated Uncaging Reactions of Propargylic Substrates,ACS Catalysis - X-MOL

Recent Developments in Metal‐Catalyzed Bio‐orthogonal Reactions for Biomolecule Tagging - Jang - 2019 - ChemBioChem - Wiley Online Library
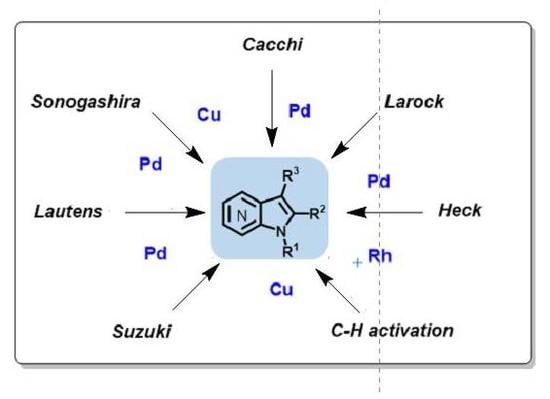
Molecules | Free Full-Text | Metal-Catalyzed Cross-Coupling Reactions on Azaindole Synthesis and Functionalization | HTML
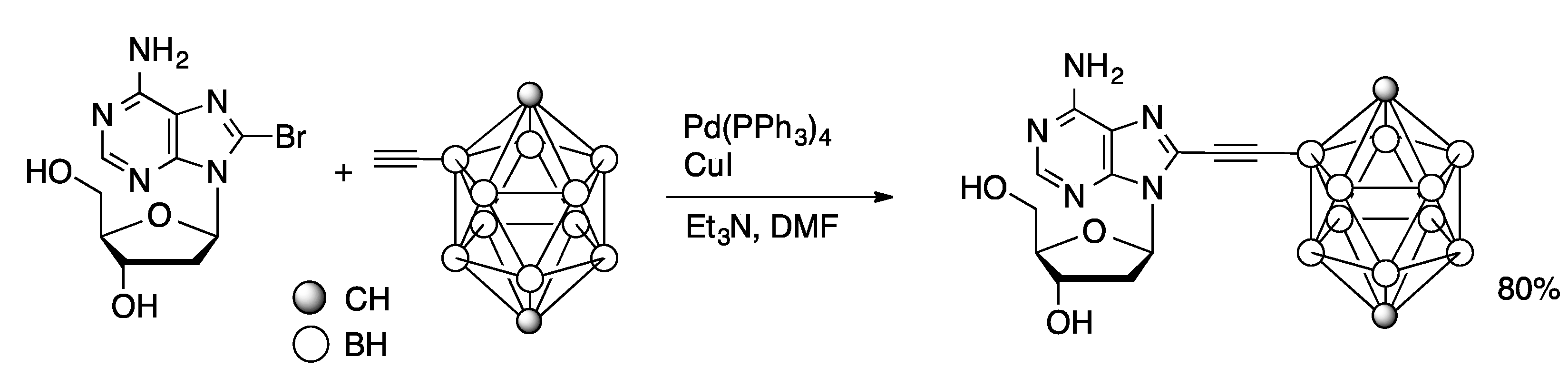
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

Synthesis of d-glyco-alkynone derivatives via carbonylative Sonogashira reaction† - RSC Adv. - X-MOL